France Regulatory Affairs Outsourcing Market Size & Outlook
Related Markets
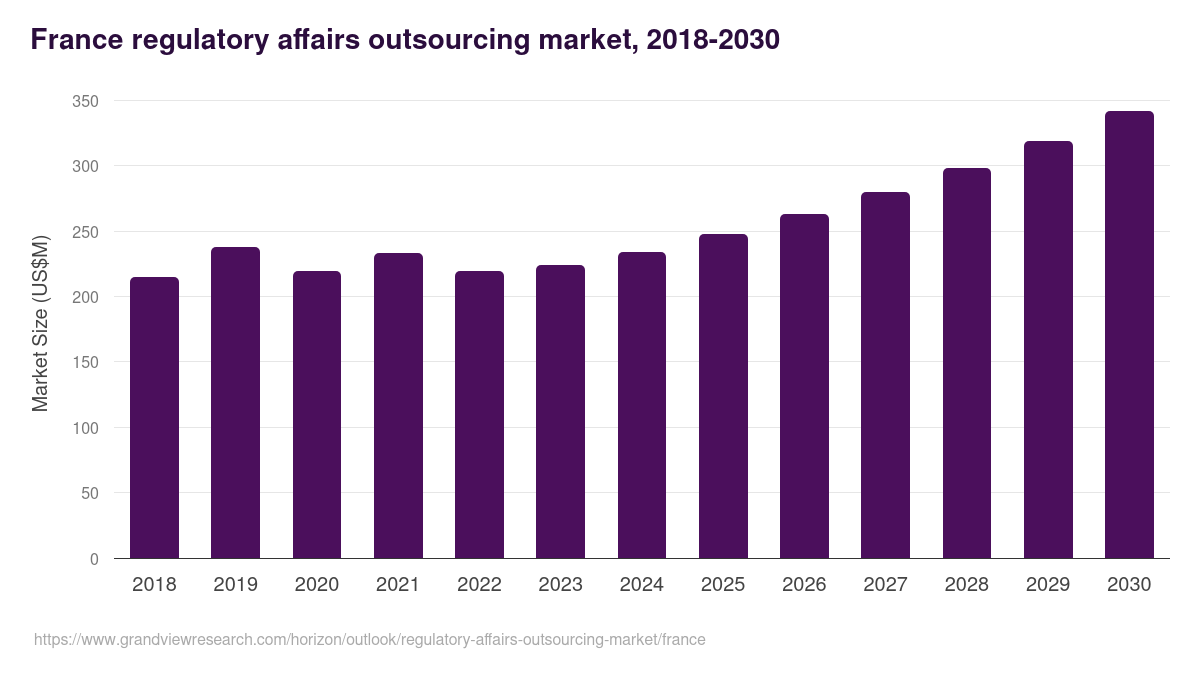
France regulatory affairs outsourcing market highlights
- The France regulatory affairs outsourcing market generated a revenue of USD 224.1 million in 2023 and is expected to reach USD 341.5 million by 2030.
- The France market is expected to grow at a CAGR of 6.2% from 2024 to 2030.
- In terms of segment, product registration & clinical trial applications was the largest revenue generating service in 2023.
- Legal representation is the most lucrative service segment registering the fastest growth during the forecast period.
Regulatory affairs outsourcing market data book summary
| Market revenue in 2023 | USD 224.1 million |
| Market revenue in 2030 | USD 341.5 million |
| Growth rate | 6.2% (CAGR from 2023 to 2030) |
| Largest segment | Product registration & clinical trial applications |
| Fastest growing segment | Legal representation |
| Historical data | 2018 - 2022 |
| Base year | 2023 |
| Forecast period | 2024 - 2030 |
| Quantitative units | Revenue in USD million |
| Market segmentation | Regulatory consulting, Legal representation, Regulatory writing & publishing, Product registration & clinical trial applications, Regulatory Submissions, Regulatory Operations, Other services |
| Key market players worldwide | Genpact Ltd, Criterium Inc., Promedica International, Wuxi AppTec Co Ltd, Medpace Holdings Inc, Charles River Laboratories International Inc, Icon PLC, Labcorp Drug Development, FREYR Battery Inc, Cencora Inc, Pharmexon, QVigilance, BlueReg Group, ZEINCRO Group |
Other key industry trends
- In terms of revenue, France accounted for 3.4% of the global regulatory affairs outsourcing market in 2023.
- Country-wise, U.S. is expected to lead the global market in terms of revenue in 2030.
- In Europe, Germany regulatory affairs outsourcing market is projected to lead the regional market in terms of revenue in 2030.
- UK is the fastest growing regional market in Europe and is projected to reach USD 426.5 million by 2030.
No credit card required*
Horizon in a snapshot
- 30K+ Global Market Reports
- 120K+ Country Reports
- 1.2M+ Market Statistics
- 200K+ Company Profiles
- Industry insights and more
Regulatory Affairs Outsourcing Market Scope
Regulatory Affairs Outsourcing Market Companies
| Name | Profile | # Employees | HQ | Website |
|---|---|---|---|---|
| ZEINCRO Group | View profile | 101-250 | Sofia, Grad Sofiya, Bulgaria, Europe | https://www.zeincro.com/ |
| BlueReg Group | View profile | 101-250 | Paris, Ile-de-France, France, Europe | https://blue-reg.com |
| QVigilance | View profile | 11-50 | Hitchin, Hertford, United Kingdom, Europe | https://www.qvigilance.com |
| Promedica International | View profile | 11-50 | Costa Mesa, California, United States, North America | https://promedica-intl.com |
| Labcorp Drug Development | View profile | 10001+ | Princeton, New Jersey, United States, North America | https://drugdevelopment.labcorp.com |
| Cencora Inc | View profile | 46000 | 1 West First Avenue, Conshohocken, PA, United States, 19428-1800 | https://www.cencora.com |
| Pharmexon | View profile | 11-50 | Prague, Hlavni mesto Praha, Czech Republic, Europe | https://pharmexon.com |
| FREYR Battery Inc | View profile | 212 | Boulevard Royal, 22-24, Level 2449, Luxembourg, Luxembourg, L-2449 | https://www.freyrbattery.com |
| Medpace Holdings Inc | View profile | 5800 | 5375 Medpace Way, Cincinnati, OH, United States, 45227 | https://www.medpace.com |
| Wuxi AppTec Co Ltd | View profile | 41116 | 288 Fute Zhong Road, Waigaoqiao Free Trade Zone, Shanghai, China, People's Republic of, 200131 | https://www.wuxiapptec.com |
| Icon PLC | View profile | 41150 | South County Business Park, Leopardstown, Dublin, Ireland, D18 X5R3 | https://www.iconplc.com |
| Criterium Inc. | View profile | 101-250 | Saratoga Springs, New York, United States, North America | http://criteriuminc.com |
| Genpact Ltd | View profile | 131000 | 22 Victoria Street, Canon's Court, Hamilton, Bermuda, HM 12 | https://www.genpact.com |
| Charles River Laboratories International Inc | View profile | 21400 | 251 Ballardvale Street, Wilmington, MA, United States, 01887 | https://www.criver.com |
France regulatory affairs outsourcing market outlook
The databook is designed to serve as a comprehensive guide to navigating this sector. The databook focuses on market statistics denoted in the form of revenue and y-o-y growth and CAGR across the globe and regions. A detailed competitive and opportunity analyses related to regulatory affairs outsourcing market will help companies and investors design strategic landscapes.
Product registration & clinical trial applications was the largest segment with a revenue share of 25.97% in 2023. Horizon Databook has segmented the France regulatory affairs outsourcing market based on regulatory consulting, legal representation, regulatory writing & publishing, product registration & clinical trial applications, regulatory submissions, regulatory operations, other services covering the revenue growth of each sub-segment from 2018 to 2030.
France is among the top eight pharmaceutical markets worldwide in terms of market size. Pharmaceutical companies have been rapidly growing in France over the last few decades. For instance, in 2018, approximately 400 drug candidates were in clinical development phase, with R&D investments of around 10.2 million.
\Such investments by these companies simultaneously boost the demand for regulatory services. France is a highly regulated market and the rate of generic penetration is low, which indirectly emphasizes the need for innovative drugs, thereby facilitating clinical trials and R&D projects in the country, which is leading to market growth.
As of 2016, France was ranked third among the EU nations and sixth across the world in terms of economy, which highlights the market attractiveness. Furthermore, supportive government initiatives such as Choose France event that showcases the country’s economic appeal & encourages foreign investments in the country are anticipated to boost the manufacturing and commercialization activities in the country, which in turn is expected to boost market growth.
Reasons to subscribe to France regulatory affairs outsourcing market databook:
-
Access to comprehensive data: Horizon Databook provides over 1 million market statistics and 20,000+ reports, offering extensive coverage across various industries and regions.
-
Informed decision making: Subscribers gain insights into market trends, customer preferences, and competitor strategies, empowering informed business decisions.
-
Cost-Effective solution: It's recognized as the world's most cost-effective market research database, offering high ROI through its vast repository of data and reports.
-
Customizable reports: Tailored reports and analytics allow companies to drill down into specific markets, demographics, or product segments, adapting to unique business needs.
-
Strategic advantage: By staying updated with the latest market intelligence, companies can stay ahead of competitors, anticipate industry shifts, and capitalize on emerging opportunities.
Target buyers of France regulatory affairs outsourcing market databook
-
Our clientele includes a mix of regulatory affairs outsourcing market companies, investment firms, advisory firms & academic institutions.
-
30% of our revenue is generated working with investment firms and helping them identify viable opportunity areas.
-
Approximately 65% of our revenue is generated working with competitive intelligence & market intelligence teams of market participants (manufacturers, service providers, etc.).
-
The rest of the revenue is generated working with academic and research not-for-profit institutes. We do our bit of pro-bono by working with these institutions at subsidized rates.
Horizon Databook provides a detailed overview of country-level data and insights on the France regulatory affairs outsourcing market , including forecasts for subscribers. This country databook contains high-level insights into France regulatory affairs outsourcing market from 2018 to 2030, including revenue numbers, major trends, and company profiles.
Partial client list
France Product Registration & Clinical Trial Applications - Regulatory Affairs Outsourcing Market, 2023 - 2030 (US$M)
France Regulatory Affairs Outsourcing Market Outlook Share, 2023 & 2030 (US$M)
Related industry reports
Related statistics
Sign up - it's easy, and free!
Sign up and get instant basic access to databook, upgrade
when ready, or enjoy our
free plan indefinitely.
Included in Horizon account
- 30K+ Global Market Reports
- 120K+ Country Reports
- 1.2M+ Market Statistics
- 200K+ Company Profiles
- Industry insights and more